As within biology, also health and pharma have various topics where science is relying on information systems to store and deliver ‘business’ critical results. As pharma science is more focused on research, health usually includes a customer factor, the patient, and information systems are set up slightly differently to accommodate this outside party.
a) Pharma
Like in the bioinformatic sector, pharma has a large data and information flow due to increased sampling and batch sized, always trying to improve or find new medical products. The range of involved sciences fields of at least chemistry, screening, preclinical and clinical bioanalysis, analytical chemistry, manufacturing R & D (Wagner, 2005). Even the fields have varying requirements on information systems, the pharma organisations tried to break functional silos and store information in one common system, which could result increased sample throughput and made it easier to ensure compliant with regulatory requirements (Thermo Fisher Scientific, 2011), it was named laboratory information management systems (LIMS). LIMS needed to run in addition to Enterprise resource planning (ERP) and customer relationship management (CRM) but data exchange to these tools was expected.
Wagner (2006) distinguishes in three kinds of LIMS:
- Custom LIMS approaches which did require huge budget for creation and maintenance and a questionable distraction from the core business, however the business process and information flow could be tailored to the business needs. With an always evolving environment, high costs were foreseeable, but could be justified.
- The “generic LIMS” was an independent software supporting various database platforms and operating systems; a high interconnection of information streams into ERP, CRM, quick deployment and easy consumption was key in this sector, for example the Accord software which is also specialized on common Excel or SQL(Accord, 2003). These generic LIMS do often lack support of all the pharma organisations core work like product-, study- or patient-orientated company tasks and surroundings like regulatory requirements. Generic LIMS can usually be slightly customized via, for example, mark-up languages but this introduces again the downsides of customized code/software.
- Commercial off-the-shelf (COTS) LIMS products do allow for information storage of these core work activities like their data housing model would cover the sample/study/MIS aspects as well as the data logging during testing and production.
This allows for one information system without extra silos for ERP or CRM databases, enabling easier data mining or business process mapping.
b) Health and Hospitals
Healthcare and especially hospitals requirements on IS are quite different, as they are more on the consumer side of the medical environment, including pharma. Given a hospital information system was build up with a hermeneutic approach as suggested by C. Winter (1994), the hospital staff is happily working with IT like tablets instead of pen and paper, breaking siloed information free for the benefit of the customer – in this case a patient.
How to establish IS in a hospital environment was described by Ahmadi, Nilashi, Shahmoradi, & Ibrahim (2017), see also figure 1 for all factors, highlighted that external environmental pressures and human dimension need to be included.
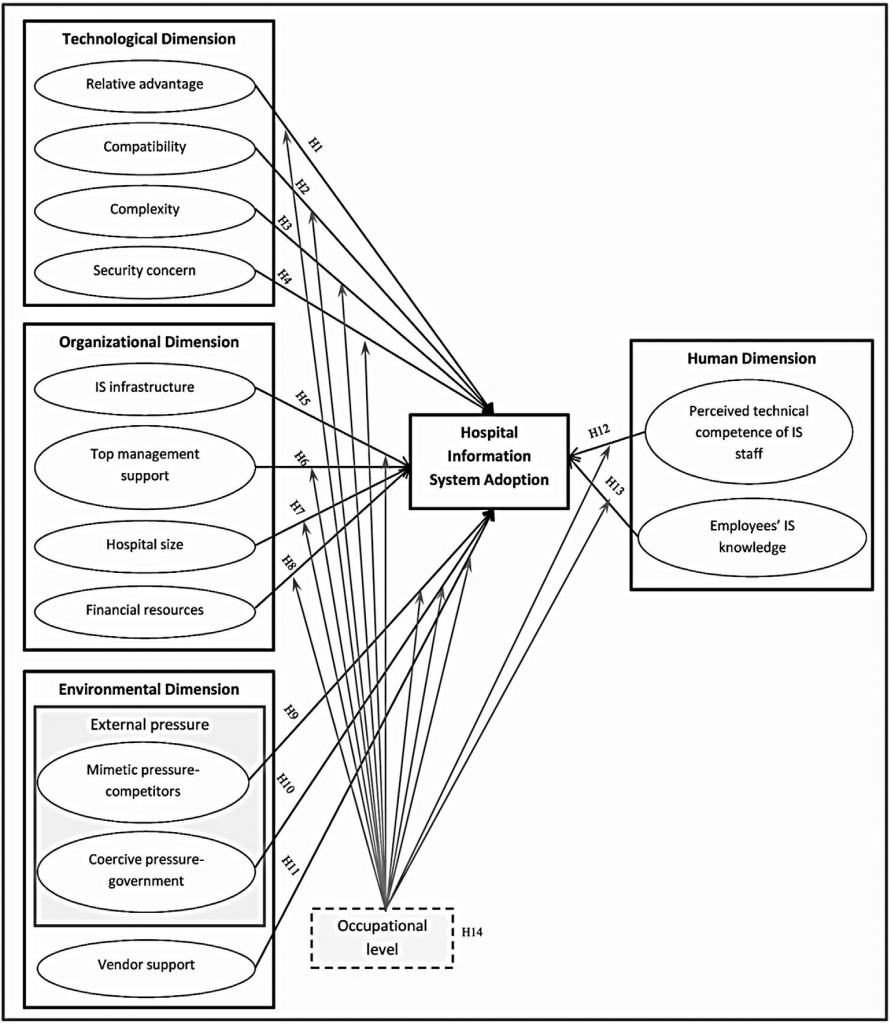
Figure1: Adoption to IS in a hospital environment and to be regarded factors
Source: Adapted from: Ahmadi, H., Nilashi, M., Shahmoradi, L., & Ibrahim, O. (2017). Hospital Information System adoption: Expert perspectives on adoption framework for Malaysian public hospitals. Computers in Human Behavior, fig.1
This establishment of IS in a hospital can then be a start for further implementations to improve services like a cloud-based supply chain in order to optimize inventory while avoiding drug shortage in order to keep costs down (NewsRX LLC, 2018).
While pharma science has long established terms (like LIMS) and community in IS, only rather recent the field of Health Information Science and Systems (HISS) was proclaimed, this should again foster the ontology of IS like data mining, image processing or user interaction in the health sector (Zhang, 2012).
References
Accord. (2003). New information management system available for pharma, biotech firms. Pharma Business Week, 28.
Ahmadi, H., Nilashi, M., Shahmoradi, L., & Ibrahim, O. (2017). Hospital Information System adoption: Expert perspectives on anadoption framework for Malaysian public hospitals. Computers in Human Behavior, 161-189.
NewsRX LLC. (2018). Reports from C.G. Kochan et al Highlight Recent Findings in Management Science (Impact of cloud-based information sharing on hospital supply chain performance: A system dynamics framework). Science Letter, 1300. Retrieved from http://go.galegroup.com.salford.idm.oclc.org/ps/i.do?p=AONE&u=salcal2&id=GALE|A524019756&v=2.1&it=r&sid=AONE&asid=efe271f2
Thermo Fisher Scientific. (2011). Pharma LIMS solution improves efficiency at Nova Biologicals. Manufacturing Chemist, 16.
Wagner, R. (2005). An overview of LIMS in the pharmaceutical industry: the pharmaceutical industry is in many ways unique. The challenges facing pharmaceutical companies, including large pharma, biotech and generics manufacturers are sometimes overwhelming. Diverse laborato. Pharmaceutical Technology Europe, p. 54+. Retrieved from http://link.galegroup.com.salford.idm.oclc.org/apps/doc/A141054959/AONE?u=salcal2&sid=AONE&xid=f0a6f63f.
Winter, C. (1994). Auswirkungen des EDV-Einsatzes auf das soziale System “Krankenhaus” / The Effects of Electronic Data Processing on the Social System “Hospital”. Zeitschrift für Gesundheitswissenschaften = Journal of public health Vol2:4, 315–32.
Zhang, Y. (2012). Welcome to Health Information Science. Health Information Science and Systems, 1. Retrieved from http://www.hissjournal.com/content/1/1/1
